Are you looking for an answer to the topic “What is the effect of temperature on the solubility of gas in liquid Class 9?“? We answer all your questions at the website Ecurrencythailand.com in category: +15 Marketing Blog Post Ideas And Topics For You. You will find the answer right below.
For all gases, as the temperature increases, solubility decreases.The solubility of gases in liquids decreases with increasing temperature. Conversely, adding heat to the solution provides thermal energy that overcomes the attractive forces between the gas and the solvent molecules, thereby decreasing the solubility of the gas; pushes the reaction in Equation 4 to the left.Hence, solubility of gas in liquid decreases with increase in temperature.

What is the effect of temperature in the solubility of gas in liquid?
The solubility of gases in liquids decreases with increasing temperature. Conversely, adding heat to the solution provides thermal energy that overcomes the attractive forces between the gas and the solvent molecules, thereby decreasing the solubility of the gas; pushes the reaction in Equation 4 to the left.
What is the effect of temperature on solubility of gas in a liquid give reason Class 12?
Hence, solubility of gas in liquid decreases with increase in temperature.
How Temperature Affects the Solubility of Gases – Experiment
Images related to the topicHow Temperature Affects the Solubility of Gases – Experiment

What is the effect of temperature on the solution of gas in liquid solution?
The volume of given mass of dissolved gas in solution also increases with increase of temperature. It becomes impossible for solvent to accommodate gaseous solute in it and gas bubbles out. Hence, with increase in temperature, the solubility of a gas in a liquid decreases.
What is the effect of temperature on solubility of liquid in liquid?
An increase in temperature puts a stress on the equilibrium condition and causes it to shift to the right. The stress is relieved because the dissolving process consumes some of the heat. Therefore, the solubility (concentration) increases with an increase in temperature.
What is the effect of temperature on solubility class 9?
Effect of the temperature on solubility
i.e. At high temperature the solubility of a solution is high so it is able to dissolve more solute, but when it is cooled, the solubility of the solution decreases and due to which the solute separate out as solid.
What is the solubility of a gas in a liquid?
The solubility of a gas in a liquid is directly proportional to the partial pressure of the gas present above the surface of liquid or solution. The most general way of using Henry’s Law is that the partial pressure of a gas above a solution is proportional to the mole fraction of the gas in the solution.
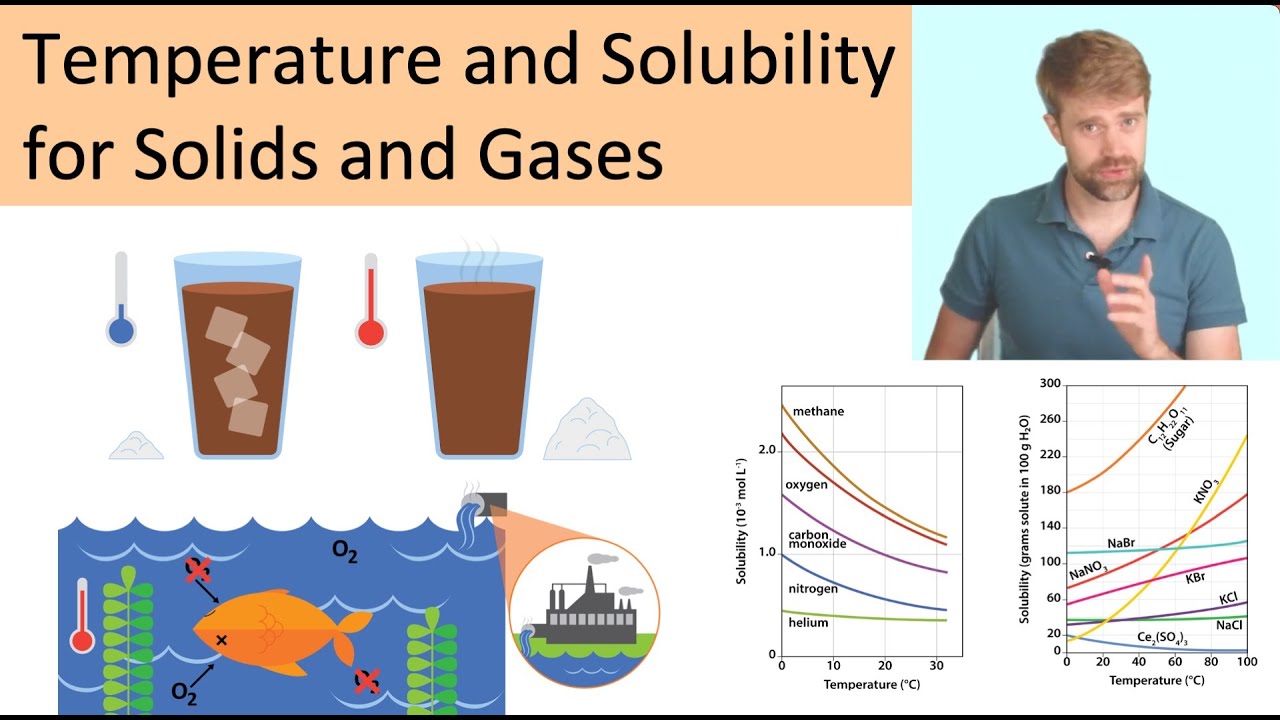
Temperature and Solubility: Solids and Gases
Images related to the topicTemperature and Solubility: Solids and Gases
See some more details on the topic What is the effect of temperature on the solubility of gas in liquid Class 9? here:
13.4: Effects of Temperature and Pressure on Solubility
The solubility of gases in liquids decreases with increasing temperature, as shown in Figure 13.4.2. Attractive intermolecular interactions in …
13.4 Effects of Temperature and Pressure on Solubility
The solubility of gases in liquids decreases with increasing temperature, as shown in Figure 13.10 “Solubilities of Several Common Gases in Water as a …
Temperature/Pressure on Solubility – Chemistry@Elmhurst
Effect of Temperature on Solubility: The solubility of solutes is dependent on temperature. When a solid dissolves in a liquid, a change in the physical …
What is the effect of temperature on the solubility of gases in liquids Class 11?
Solubility of gases in liquids decreases with increase in temperature.
What is the effect of temperature on solubility class 12?
Hence, we can say that increasing the temperature increases the solubility of solids and liquids, whereas in case of gases increase in temperature decreases the solubility of any substance.
What is the effect of temperature on solubility class 6?
The solubility of a substance in water increases on increasing the temperature. Larger amount of a substance can be dissolved in a given amount of water on heating it. The solubility of a substance decreases on lowering the temperature.
How temperature and pressure affect the solubility of gases in liquids?
Solution : Solubility of gas decreases with increase in temperature and increases with increase in pressure.
Temperature and Gas Solubility
Images related to the topicTemperature and Gas Solubility

Why does the solubility of gases in liquids decreases with increasing temperature?
As the kinetic energy of the gaseous solute increases, its molecules have a greater tendency to escape the attraction of the solvent molecules and return to the gas phase. Therefore, the solubility of a gas decreases as the temperature increases.
What are the effect of temperature and pressure on solubility of a gas in solution?
The solubility of a solid may increase or decrease with increasing temperature, whereas the solubility of a gas decreases with an increase in temperature and a decrease in pressure.
Related searches to What is the effect of temperature on the solubility of gas in liquid Class 9?
- at a particular temperature, as the pressure increases the solubility of a gas in water will
- what is the effect of temperature on the solubility of gases in liquids
- as the temperature rises the solubility of co (g) in water
- what is the effect of pressure on the solubility of gases? solids?
- what is the effect of pressure on the solubility of gases in liquids?
- solubility of gas in liquid decreases with the rise in temperature
Information related to the topic What is the effect of temperature on the solubility of gas in liquid Class 9?
Here are the search results of the thread What is the effect of temperature on the solubility of gas in liquid Class 9? from Bing. You can read more if you want.
You have just come across an article on the topic What is the effect of temperature on the solubility of gas in liquid Class 9?. If you found this article useful, please share it. Thank you very much.