Are you looking for an answer to the topic “Which electrode is the anode?“? We answer all your questions at the website Ecurrencythailand.com in category: +15 Marketing Blog Post Ideas And Topics For You. You will find the answer right below.
The Anode is the negative or reducing electrode that releases electrons to the external circuit and oxidizes during and electrochemical reaction. The Cathode is the positive or oxidizing electrode that acquires electrons from the external circuit and is reduced during the electrochemical reaction.The electrode at which oxidation takes place is known as the anode, while the electrode at which reduction take place is called the cathode. If you see galvanic cell reduction take place at the left electrode, so the left one is the cathode. Oxidation takes place at the right electrode, so the right one is the anode.The anode is the electrode where electricity moves into. The cathode is the electrode where electricity is given out or flows out. The anode is usually the positive side. A cathode is a negative side.
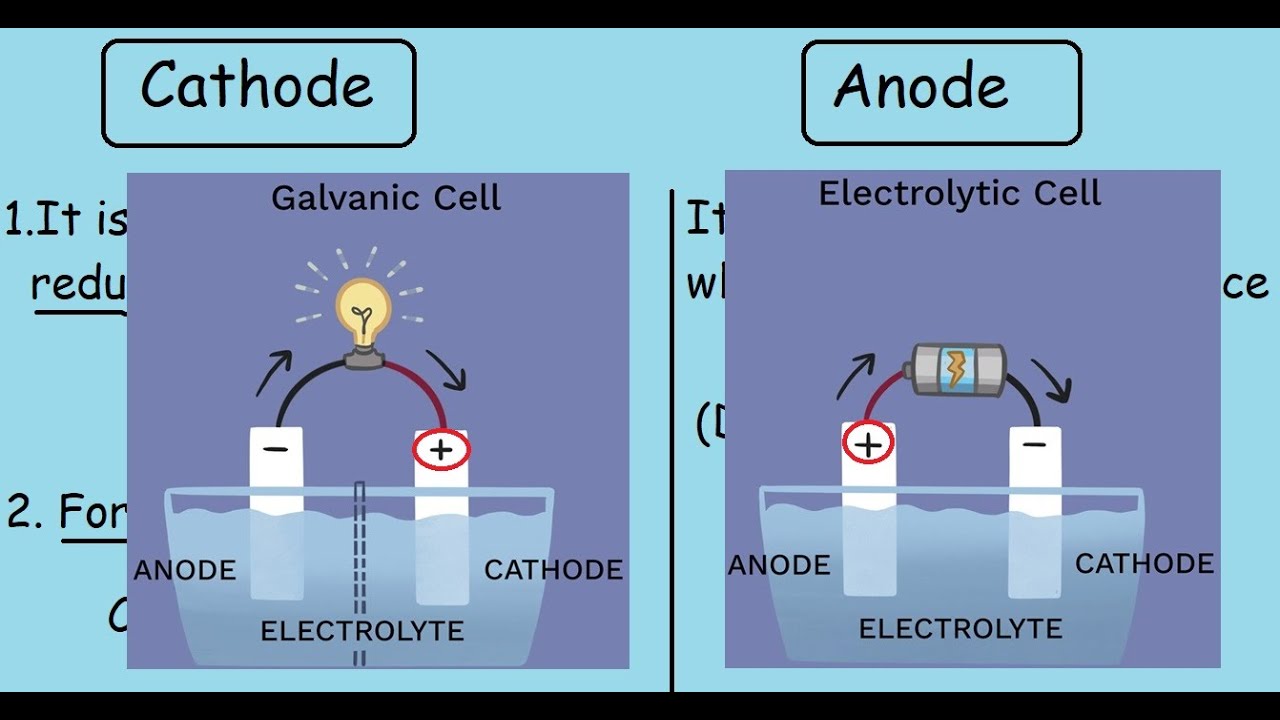
How can you tell which electrode is the anode?
The electrode at which oxidation takes place is known as the anode, while the electrode at which reduction take place is called the cathode. If you see galvanic cell reduction take place at the left electrode, so the left one is the cathode. Oxidation takes place at the right electrode, so the right one is the anode.
How do you know which electrode is anode or cathode?
The anode is the electrode where electricity moves into. The cathode is the electrode where electricity is given out or flows out. The anode is usually the positive side. A cathode is a negative side.
Cathode and Anode |Quick differences and comparisons|
Images related to the topicCathode and Anode |Quick differences and comparisons|

Which electrode is positive anode or cathode?
During charge, the positive electrode is an anode, and the negative electrode is a cathode.
Which electrode is called the cathode?
The cathode is the negatively charged electrode. The cathode attracts cations or positive charge. The cathode is the source of electrons or an electron donor.
Is the anode always on the left?
The anode is always placed on the left side, and the cathode is placed on the right side. The salt bridge is represented by double vertical lines (||).
Is anode positive or negative electrode?
anode, the terminal or electrode from which electrons leave a system. In a battery or other source of direct current the anode is the negative terminal, but in a passive load it is the positive terminal.
Why is the anode negative?
Anode is negative in electrochemical cell because it has a negative potential with respect to the solution while anode is positive in electrolytic cell because it is connected to positive terminal of the battery.
See some more details on the topic Which electrode is the anode? here:
Anode – Wikipedia
An anode is an electrode through which the conventional current enters into a polarized electrical device. This contrasts with a cathode, an electrode …
What is the difference between positive and negative …
An anode is an electrode where an oxidation reaction occurs (loss of electrons for the electroactive species). · A cathode is an electrode where …
How to Define Anode and Cathode – ThoughtCo
The anode is the positively charged electrode. · The anode attracts electrons or anions. · The anode may be a source of positive charge or an …
Definition and Difference Between Anode and Cathode
A Cathode is a negative electrode, whereas the anode is a positive electrode. They are so-called because the cations, which are positively charged, migrate to …
Is anode always positive?
In a battery or galvanic cell, the anode is the negative electrode from which electrons flow out towards the external part of the circuit.
What is a negative electrode?
The negatively charged electrode in electrolysis is called the cathode . Positively charged ions move towards the cathode. The positively charged electrode in electrolysis is called the anode . Negatively charged ions move towards the anode.
What is the charge on cathode?
A cathode is a negatively charged electrode. However, the polarity of the cathode with respect to anode can either be negative or positive, and it largely depends on the operation of the device. For example, in a recharging battery the cathode is negative.
What Is Electrolysis | Reactions | Chemistry | FuseSchool
Images related to the topicWhat Is Electrolysis | Reactions | Chemistry | FuseSchool

Is cathode positive or negative in diode?
Diode Polarity & Symbols
One side is the positive terminal, called the anode. The other terminal is the negative end, called the cathode. Going back to our flow of electricity, current can only move in a diode from the anode to the cathode, never the other way around.
What is positive electrode?
(ăn′ōd′) 1. The positive electrode in an electrolytic cell, toward which negatively charged particles are attracted. The anode has a positive charge because it is connected to the positively charged end of an external power supply.
Why is the cathode positive?
A battery or galvanic cell in use has a cathode that is the positive terminal since that is where conventional current flows out of the device. This outward current is carried internally by positive ions moving from the electrolyte to the positive cathode (chemical energy is responsible for this “uphill” motion).
What is the positive electrode called in electrolysis?
The positively charged electrode in electrolysis is called the anode .
Is cathode positive or negative in electrolysis?
1: An electrolytic cell. The battery pumps electrons away from the anode (making it positive) and into the cathode (making it negative).
What happens at the cathode?
The reaction at the anode is oxidation and that at the cathode is reduction. The electrons are supplied by the species getting oxidized. They move from anode to the cathode in the external circuit.
Why anode is positive and cathode is negative in electrolytic cell?
In an electrolytic cell, the cathode is considered to be negative and the anode is positive because the battery pumps electrons aways from the anode (which makes anode positive) into the cathode (making the cathode negative).
Is anode negatively charged?
Charge on Anode and Cathode
At the anode, there is an oxidation response. The oxidized species would lose the electrons, leaving this electrode with an electron accumulation. Thus, the anode is negatively charged.
5.0.2 Use the terms electrode, electrolyte, anode and cathode
Images related to the topic5.0.2 Use the terms electrode, electrolyte, anode and cathode

How can you tell which electrode is negatively charged?
The electrode at which oxidation takes place, behaves as anode and it is negatively charged. Hence zinc electrode is negatively charged. Was this answer helpful?
What is the symbol of cathode?
K = Cathode (because “C” was reserved for Collector)
Related searches to Which electrode is the anode?
- anode is positive or negative
- what is an anode
- what is electrode
- which gas is produced at the positive electrode (anode)
- what is anode and cathode
- which electrode is the anode in a galvanic cell
- anode symbol
- anode and cathode metals
- which electrode is the anode in a galvanic cell apex
- anode and cathode charges
- which electrode is the anode and which is the cathode
- which electrode is the anode chegg
- which electrode is the anode
- anode and cathode sign
- how do you know which electrode is the anode
- which electrode anode or cathode is the oxidising electrode why
- the anode is the electrode at which oxidation takes place
- how to determine which electrode is the anode
Information related to the topic Which electrode is the anode?
Here are the search results of the thread Which electrode is the anode? from Bing. You can read more if you want.
You have just come across an article on the topic Which electrode is the anode?. If you found this article useful, please share it. Thank you very much.